BACKGROUND: Relapsed/refractory (R/R) B-cell non-Hodgkin lymphoma (B-NHL) remains an area of high unmet patient need and no curative options are currently available.
Odronextamab (REGN1979) is a first-in-class, hinge-stabilized, fully human IgG4-based bispecific antibody that binds to CD20-expressing cells and CD3 on T cells, targeting CD20+ cells via T-cell-mediated cytotoxicity independent of T-cell receptor recognition. The safety, tolerability, and anti-tumor activity of odronextamab monotherapy was evaluated in a global, multicenter, Phase 1 study in heavily pretreated patients with R/R B-NHL (NCT02990951; Bannerji et al, ASH 2019). Intravenous infusion of odronextamab has demonstrated an acceptable safety profile at doses up to 320 mg weekly (QW), and the maximum tolerated dose was not reached. Broad and durable anti-tumor responses were observed in both indolent and aggressive lymphomas, including in patients who progressed after prior CAR T-cell therapy. An assessment of pharmacokinetics, efficacy and safety data from the Phase 1 study informed the recommended Phase 2 dosing regimens.
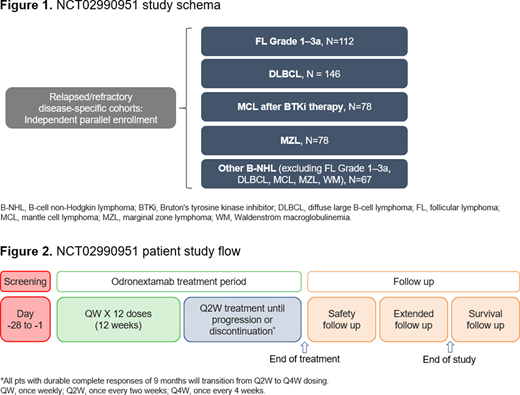
METHODS: This global, Phase 2, open-label, multi-cohort study (R1979-ONC-1625; NCT03888105) is designed to assess the anti-tumor activity and safety of odronextamab in patients with B-NHL. There are five disease-specific cohorts, each with independent parallel enrollment. The study includes patients with: (1) R/R follicular lymphoma (FL) Grade 1-3a after ≥2 prior lines of systemic therapy, including an anti-CD20 antibody and an alkylating agent; (2) R/R diffuse large B-cell lymphoma (DLBCL) after ≥2 lines of systemic therapy, including an anti-CD20 antibody and an alkylating agent; (3) R/R mantle cell lymphoma (MCL) following or with failure to tolerate Bruton's tyrosine kinase inhibitor therapy; (4) R/R marginal zone lymphoma (MZL) after ≥2 lines of systemic therapy; (5) other R/R B-NHL subtypes, excluding Waldenström macroglobulinemia, after ≥2 lines of systemic therapy (Fig. 1).
Estimated total enrollment is 481 patients. Key eligibility criteria are: age ≥18 years; not appropriate for other approved therapy with established benefit; ≥1 bi-dimensionally measurable nodal lesion of ≥1.5 cm; Eastern Cooperative Oncology Group performance status ≤1; and adequate bone marrow, renal, and hepatic function. Key exclusion criteria are: prior anti-CD20 x CD3 bispecific antibody therapy; prior CAR T-cell therapy; primary central nervous system (CNS) lymphoma or known involvement by non-primary CNS NHL; and history of allogeneic hematopoietic stem cell transplantation.
Odronextamab is administered using a step-up dose schedule consisting of an initial dose at Week (W)1, an intermediate dose at W2, and thereafter, a fixed weekly dose until W12 followed by maintenance Q2W dosing until progression or discontinuation (Fig. 2). The dose for indolent B-NHL is 80 mg QW followed by 160 mg Q2W, and for aggressive B-NHL is 160 mg QW followed by 320 mg Q2W. All patients with durable complete responses of 9 months will transition from Q2W to Q4W dosing.
The primary endpoint for each cohort is objective response rate (ORR) by independent central review, as assessed from first dose until completion of 28 weeks of study treatment, or study withdrawal. Secondary endpoints include complete response (CR) rate, progression-free survival, duration of response, disease control rate (DCR), overall survival, incidence and severity of treatment-emergent adverse events, pharmacokinetics, immunogenicity, and patient-reported outcomes.
ORR, CR rate and DCR with a two-sided 95% confidence interval (CI) will be summarized. Time-to-event endpoints will be summarized by median and corresponding 95% CI using the Kaplan-Meier method.
The study is actively accruing patients at sites across North America, Europe, and Asia-Pacific.
Kim:AstraZeneca: Consultancy; Novartis: Consultancy; Takeda: Consultancy; Sanofi: Consultancy; F. Hoffmann-La Roche Ltd/Genentech, Inc.: Consultancy; Voronoi: Consultancy; Boryung: Consultancy; AstraZeneca and Korea Health Industry Development Institute: Research Funding. Stevens:Amgen, MorphoSys: Consultancy. Poon:Astrazeneca, Pfizer, Takeda, Janssen, Roche, Novartis: Honoraria. Le Gouill:Loxo Oncology at Lilly: Consultancy; Roche Genentech, Janssen-Cilag and Abbvie, Celgene, Jazz pharmaceutical, Gilead-kite, Loxo, Daiichi-Sankyo and Servier: Honoraria. Carpio:Takeda, Regeneron: Consultancy. Keating:Celgene: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Taiho: Membership on an entity's Board of Directors or advisory committees; Shire: Membership on an entity's Board of Directors or advisory committees; Servier: Membership on an entity's Board of Directors or advisory committees; Seattle Genetics: Consultancy; Sanofi: Membership on an entity's Board of Directors or advisory committees; Merck: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees; Hoffman La Roche: Membership on an entity's Board of Directors or advisory committees; Takeda: Honoraria, Membership on an entity's Board of Directors or advisory committees; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding. Adriaens:Regeneron Pharmaceuticals, Inc: Current Employment, Current equity holder in publicly-traded company, Patents & Royalties: Dosing regime that mitigates cytokine release syndrome for therapeutic antibodies (status: pending). Ufkin:Regeneron Pharmaceuticals, Inc: Current Employment, Current equity holder in publicly-traded company. Sabir:Regeneron Pharmaceuticals, Inc: Current Employment, Current equity holder in publicly-traded company. Li:Regeneron Pharmaceuticals, Inc: Current Employment, Current equity holder in publicly-traded company. Jankovic:Regeneron Pharmaceuticals, Inc: Current Employment, Current equity holder in publicly-traded company. Zhu:Regeneron Pharmaceuticals, Inc: Current Employment, Current equity holder in publicly-traded company. Brouwer-Visser:Regeneron Pharmaceuticals, Inc: Current Employment, Current equity holder in publicly-traded company. Leng:Regeneron Pharmaceuticals, Inc: Current Employment, Current equity holder in publicly-traded company. Sirulnik:Regeneron Pharmaceuticals, Inc.: Current Employment, Current equity holder in publicly-traded company. Chaudhry:Regeneron Pharmaceuticals, Inc: Current Employment, Current equity holder in publicly-traded company. Ambati:Regeneron Pharmaceuticals, Inc: Current Employment, Current equity holder in publicly-traded company. Won Seog:Roche, Takeda, J&J, Kyowa-Kirin, Celltrion ,Pfizer, Donga: Research Funding.
The Trial in Progress abstract will report on use of odronextamab in a Phase 2 clinical trial of patients with B-NHL
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal